Improved electrochemical performance of Na2/3Ni1/2Mn1/2O2 through stabilizing the oxygen redox activity
M. Kalapsazova*, P.Markov, R. Kukeva, E. Zhecheva* and R. Stoyanova*

Abstract:
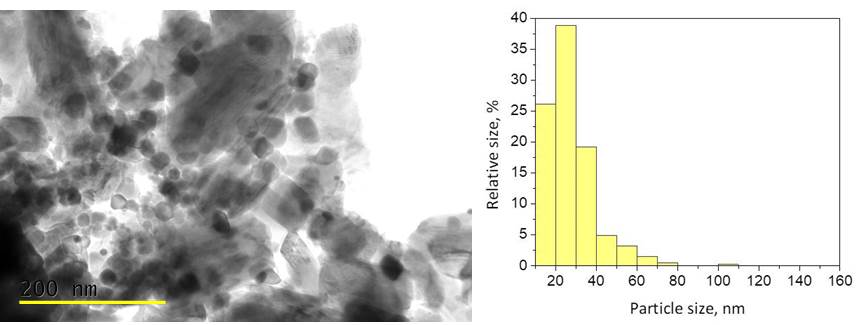
The ever-growing renewable energy storage industry imposes great demands that the current most efficient storage devices, lithium-ion batteries (LIBs), are unable to meet. Among all post lithium-ion battery approaches, the sodium-ion technology (SIBs) apparently is the most interesting. Unlike lithium, sodium is an abundant and cheaper element. Although SIBs provide similar performance, the current drawbacks of sodium-ion batteries are the limited number of cycles and energy storage densities. The shortest route to increase the energy density of sodium-ion batteries is through the simultaneous activation of transition metal and oxygen redox reactions (i.e. TMm+/TMn+ and O2−/O2n−) at intercalation-type electrode materials. Herein, we propose a new concept to stabilize oxygen redox activity by using oxygen-storage materials as efficient buffer supplying and receiving an oxygen during alkali-ion intercalation. The concept consists in treatment of intercalation oxides, P3-Na2/3Ni1/2Mn1/2O2, with “oxygen-storage” material, namely CeO2. In the mixture between P3-Na2/3Ni1/2Mn1/2O2 and CeO2 phases, there is a close particles contact, which is developed on a nano-scale range. The CeO2 suppresses the particle aggregation of the layered oxide and particles with dimensions between 10 and 40 nm become dominant (Fig.1). This type of modification with CeO2 does not affect the bulk structure of layered oxide.
The modified oxides display drastic increase in the reversible capacity when used as cathodes in Na–, Li– and hybrid Li,Na-ion cells. The highest capacity of about 285 mAh/g is reached for the modified oxide cycled in hybrid Li,Na-ion cell. The reversible capacity increases following the order Na+ intercalation < Li+ intercalation < Li+,Na+ co-intercalation (Fig.2). On the one hand, this indicates a synergistic effect of the dual Li+,Na+ intercalation in order to reach a higher capacity. On the other, irrespective of the type of alkali ion intercalation the higher intercalation capacities of the modified oxide are in agreement with the concept of the beneficial effect of CeO2 on oxygen-redox reaction. It is worth mentioning that CeO2 is not participating in the electrochemical reaction, according to the ex-situ HR-TEM.

In addition, CeO2 affects also the inter-phase interaction between electrode and Li/NaPF6 salt electrolyte. These data could serve as guidelines to achieve colossal reversible capacity at layered lithium and sodium transition metal oxides – research dream from the beginning until now in the development of lithium-ion batteries.
The authors from CARiM’s Research Team are noted with *
