Mechanochemically induced transformation of NaFePO4 maricite: anion redox activity and high specific capacity
T. Boyadzhieva, V. Koleva* and R. Stoyanova*
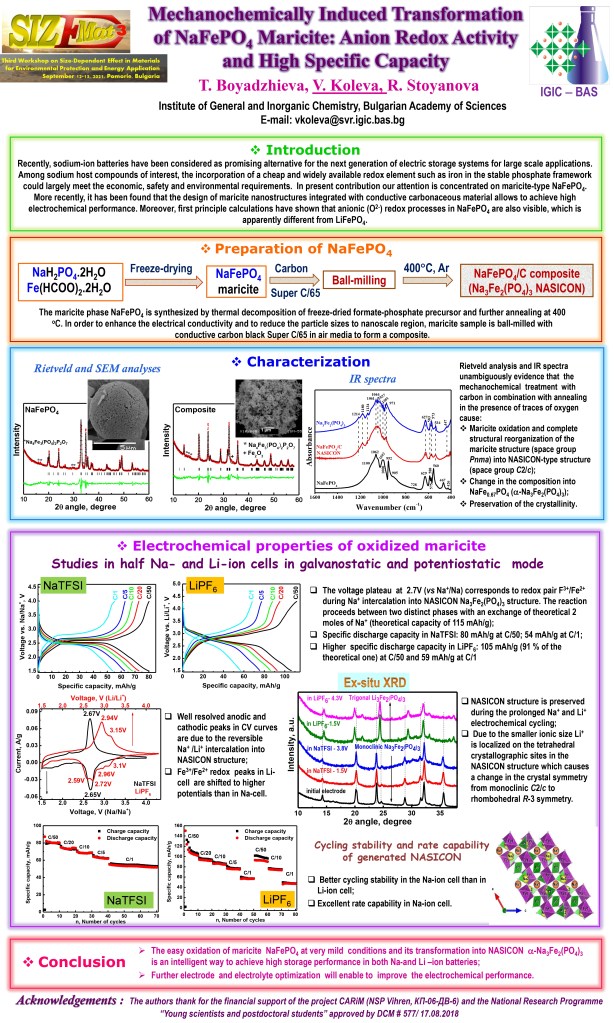
The rapid depletion of lithium as a key chemical element in the lithium-ion technology and the continuous increase in its cost have posed the urgent task of researchers to develop alternative batteries such as Na-ion batteries based on more abundant, readily available, inexpensive and environmentally-friendly electrode materials. The commercialization of the Na-ion battery, however, is still a great challenge and one of the important issues is attributed to the choice of the most suitable electrode materials. Among sodium–host compounds of interest, the incorporation of a cheap and widely available redox element such as iron in the stable phosphate framework could largely meet the economic, safety and environmental requirements. In present contribution our attention is concentrated on maricite-type NaFePO4 (M-NFP). More recently, it has been found that the design of maricite nanostructures integrated with conductive carbonaceous material allows to achieve high electrochemical performance. Moreover, first principle calculations have shown that anionic (O2-) redox processes in NaFePO4 are also visible, which is apparently different from LiFePO4. The maricite phase NaFePO4 is synthesized by thermal decomposition of freeze-dried formate-phosphate precursor and further annealing at 400 °C. In order to enhance the electrical conductivity and to reduce the particle sizes to nanoscale region, M-NFP is ball-milled with conductive carbon black Super C/65 in air media. The electrochemical properties of the as-formed composite M-NFP/C are studied in half sodium and lithium ion cells using NaTFSI and LiPF6 electrolytes in galvanostatic and potensiostatic mode at different charge/discharge and scan rates. The mechanochemical treatment is found to cause a complete structural reorganization and the initial maricite structure (space group Pnma) is fully transformed into NASICON-type structure (space group C2/c) with change in the composition into NaFe0.67PO4 (Na3Fe2(PO4)3) and preservation of the crystallinity (Fig. 1A-D). The cyclic voltammograms (CV) of the as-formed NASICON phase evidence that Na+ and Li+ intercalation proceeds with participation of the Fe2+/Fe3+ ionic pair, but in a different way: Na+ intercalation is one step process in contrast to the consecutive Li+ intercalation (Fig. 1E).
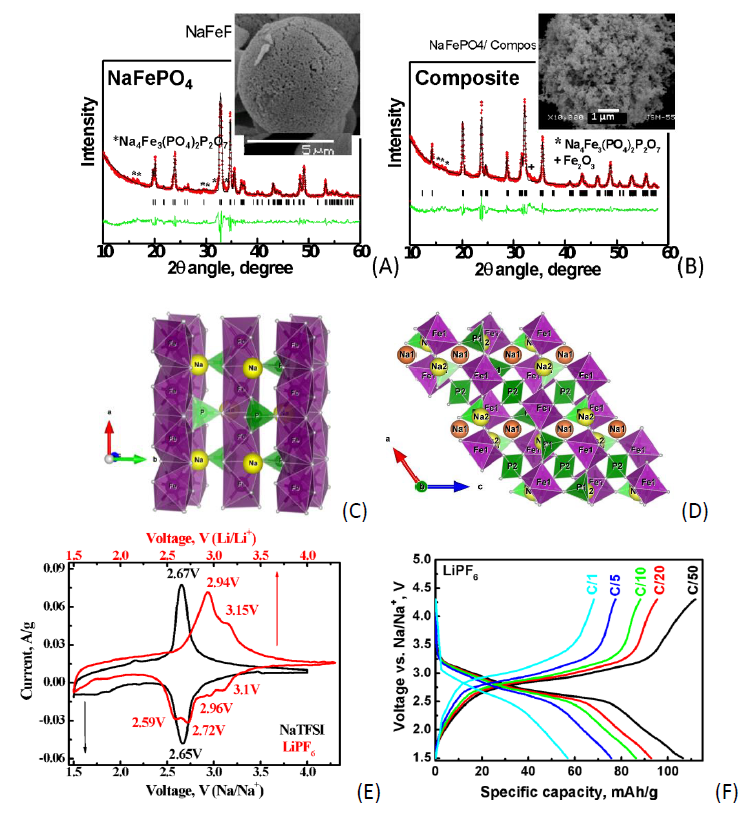
The as-generated nanosized NASICON phase Na3Fe2(PO4)3 exhibits good storage performance in both Na- and Li-ion cells. The specific discharge capacity in LiPF6 at C/50 is 112 mAh/g (97 % of the theoretical one) and 59 mAh/g at C/1 rate (Fig. 1F). Ex-situ XRD measurements reveal that the NASICON structure is preserved during the prolonged Na+ and Li+ electrochemical cycling. However, due to the smaller ionic size Li+ is localized on tetrahedral crystallographic sites in the NASICON structure which causes a change in the crystal symmetry: from monoclinic C2/c to rhombohedral R-3 symmetry. The obtained results clearly manifest that the mechanochemically generated NASICON Na3Fe2(PO4)3 phase has a great potential as a positive electrode for Na-ion batteries.
The authors from CARiM’s Research Team are noted with *
