Enhancing oxygen redox activity in sodium-deficient three-layered oxides: modeling the structure or the surface?
Mariya Kalapsazova *, Rositsa Kukeva, Ekaterina Zhecheva * and Radostina Stoyanova *
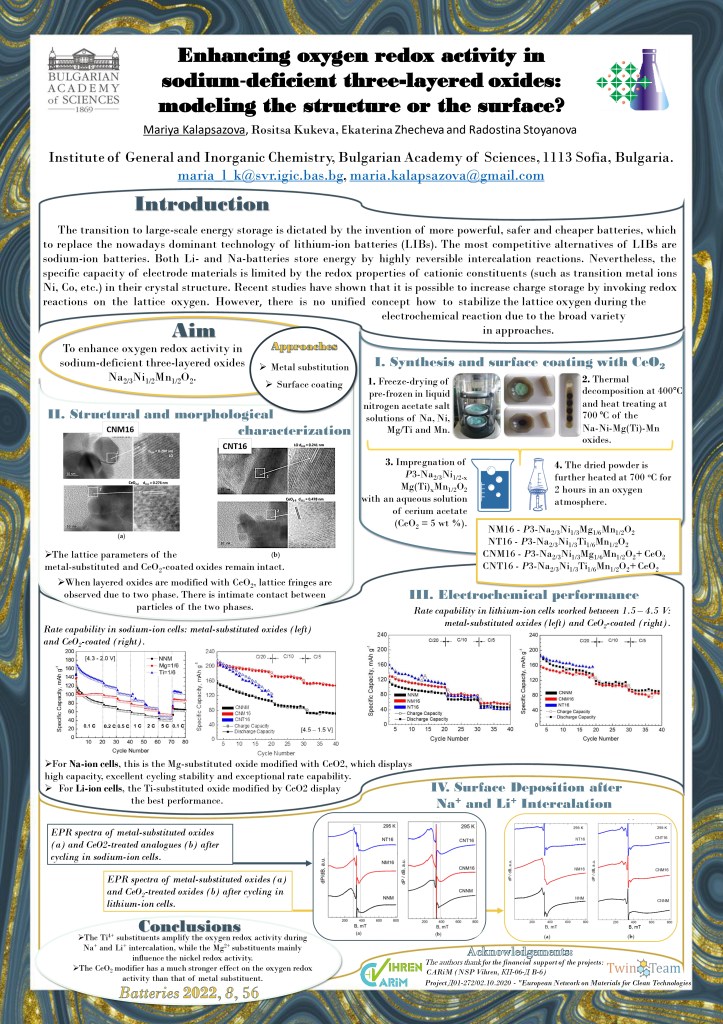
Abstract: The transition to large-scale energy storage is dictated by the invention of more powerful, safer and cheaper batteries, which to replace the nowadays dominant technology of lithium-ion batteries (LIBs). The most competitive alternatives of LIBs are sodium-ion batteries. Both Li- and Na-batteries store energy by highly reversible intercalation reactions. Nevertheless, the specific capacity of electrode materials is limited by the redox properties of cationic constituents (such as transition metal ions Ni, Co, etc.) in their crystal structure. Recent studies have shown that it is possible to increase charge storage by invoking redox reactions on the lattice oxygen [1,2]. However, there is no unified concept how to stabilize the lattice oxygen during the electrochemical reaction due to the broad variety in approaches.
Herein, we unveil the interplay between metal substitution, treatment with oxygen storage materials and anionic redox activity in sodium nickel manganese oxides with three-layer stacking (P3-Na2/3Ni1/2Mn1/2O2) during single Na+ and Li+ intercalation. As metal substituents, Mg2+ and Ti4+ ions were used through partial replacement of low- and high-oxidized nickel ions, respectively: Na2/3Ni1/3Mg1/6Mn1/2O2 and Na2/3Ni1/3Ti1/6Mn1/2O2. Furthermore, the metal-substituted oxides are
treated with an oxygen storage material, such as CeO2.

The effect of the metal substitution and CeO2-treatment on the structure and morphology of the oxides is observed by X-ray powder diffraction, EPR spectroscopy and HR-TEM. Cyclic voltammetry was employed to investigate the reduction and oxidation processes during Na+ and Li+ intercalation. The electrochemical tests are carried out in galvanostatic mode. The electrode–electrolyte interaction is monitored by ex-situ EPR of the long-cycled electrodes.
The Ti4+ substituents amplify the oxygen redox activity during Na+ (Fig. 1; (C)NT16) and Li+ intercalation (Fig.2), thus permitting reaching the highest specific capacity at potentials above 4.2 V. At the same time, the interaction of oxides with NaPF6– and LiPF6-based electrolytes is intensified, leading to a deposition of MnF2. As a result, Ti-substituted oxide displays moderate cycling stability and poor rate capability. Contrary to Ti4+, the Mg2+ substituents mainly influence the nickel redox activity and suppress the deposition of MnF2, all of them contributing to the exceptional cycling stability and rate capability (Fig.1,2; (C)NM16). The CeO2 modifier has a much stronger effect on the oxygen redox activity than that of metal substituents; thus, the highest specific capacity is attained (Fig.1,2 right). In addition, the CeO2 modifier tunes the electrode–electrode interaction by eliminating the deposition of MnF2.

coated (right).
By combining the appropriate substituents with the CeO2 modifier, the best electrode material was selected. For Na-ion cells, this is the Mg-substituted oxide modified with CeO2, which displays high capacity, excellent cycling stability and exceptional rate capability. For Li-ion cells, the Ti-substituted oxide modified by CeO2 display the best performance.
References
1. M. M. Rahman and F. Lin, Matter 4 (2021) 490–527.
2. M. Kalapsazova, K. Kostov, R. Kukeva, E. Zhecheva and R. Stoyanova, J. Phys. Chem. Lett. 12, 32
(2021) 7804–7811.
The authors from CARiM’s Research Team are noted with *
