Authors: Mariya Kalapsazova, Krassimir Kostov, Rositsa Kukeva, Ekaterina Zhecheva, and Radostina Stoyanova

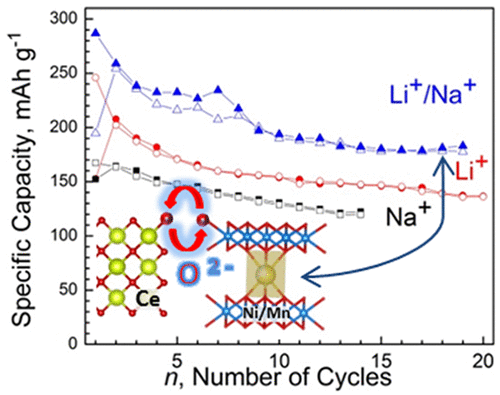
Abstract: To double the energy density of lithium- and sodium-ion batteries there is a need to activate simultaneously cationic and anionic redox reactions at the intercalation-type electrodes. In contrast to the cationic redox activity, the oxygen redox activity enforces an enhancement in the surface reactivity of the oxides leading to their poor reversibility and cycling stability. Herein, we propose a new concept to stabilize oxygen redox activity by using oxygen-storage materials as an efficient buffer supplying and receiving oxygen during alkali ion intercalation. As a proof-of-concept, the study is focused on CeO2 as a modifier of sodium nickel–manganese oxide with a three-layer sequence, P3-Na2/3Ni1/2Mn1/2O2. TheCeO2-modifiedP3-Na2/3Ni1/2Mn1/2O2 displays a drastic increase in the reversible capacity following the order Na+ intercalation < Li+ intercalation < Li+,Na+ cointercalation.
