Effect of Ce modification on the electrochemical properties of NaFexPO4 as an electrode material for sodium-ion batteries
Violeta Koleva, Trajche Tushev, Sonya Harizanova, Radostina Stoyanova
Abstract:
1. Introduction – Sodium-ion batteries exhibit lower energy density than lithium-ion batteries, but they are particularly promising for stationary storage where low cost, long life, safety and sustainability are of primary importance. The selection of the most suitable electrode materials is still a serious challenge that hinders the commercialization of the Na-ion batteries as a competitive alternative to Li-ion batteries. In this regard sodium iron phosphates exhibiting variety of compositions and structures (NaxFey(PO4)z) have attracted increasing attention as promising intercalation electrode materials due to their low cost, safety and being environmental friendly. Intercalation-based cathodes typically rely on the cationic redox activity of transition metals. However, the activation of both cationic and anionic redox reactions in the intercalation-type electrodes has been found to be an effective way to increase drastically the reversible capacity of P3-Na2/3Ni1/2Mn1/2O2 by using CeO2 as an oxygen storage material. Moreover, first principle calculations have shown that anionic (O2-) redox processes in NaFePO4 are also visible . Inspired by these data the main idea of present contribution is to study effect of Ce-modification on electrochemical properties of NaFexPO4 as cathode material in sodium and lithium half-cells.
2. Experimental – Ce-modified NaFexPO4 materials (denoted NFP-2Ce and NFP-5Ce, containing about
1 and 2 % Ce, respectively) were prepared by a freeze-drying method. The electrochemical properties were studied in galvanostatic and potentiostatic regimes in sodium and lithium half-cells at 20 and 40 °C. The cycling stability was tested with a C/2 rate.
3. Results and Discussion – The presence of Ce ions affects the phase composition of NaFexPO4: it
comprises a mixture of NASICON and alluaudite-type phosphate. Well resolved anodic and cathodic
peaks in cyclic voltammograms (CV) are attributed to the Na+ (Li+) intercalation into NASICON and
Alluaudute structures owing to Fe3+/Fe2+ ion pairs (Image 1a,b).
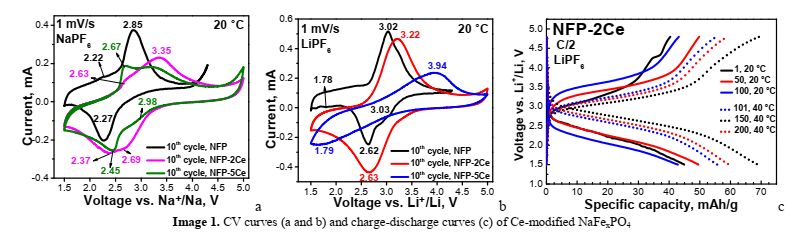
The Fe3+/Fe2+ redox potentials in Li-cells are shifted to higher potentials than in Na-cells. Ce ions are electrochemically inactive, but their presence leads to several effects: (i) progressive increase in the redox potentials in the two ion cells; (ii) redox peaks broadening and (iii) increase in peak-to-peak separation (suppression in the electron transfer reactions). The Cen+ ions provoke strong polarization of the ion cells, more pronounced in Li half-cells, and this causes capacity decrease during the cycling (Image 1c).
4. Conclusions – Further electrode optimization will enable to overcome the strong polarization and improve the electrochemical performance. More studies are needed to clarify the state of Cen+ ions in the electrodes and their role to affect the electrochemical behavior of the NaFexPO4 electrodes.
Acknowledgements: The authors thank to the financial support of project “CARiM (grant number № КП-06-ДB-6) and project “Energy Storage and Hydrogen Energy” (grant number № Д01-349/13.12.2023) for incubators used.

